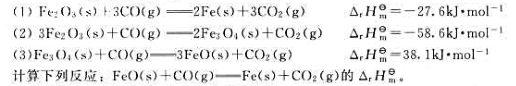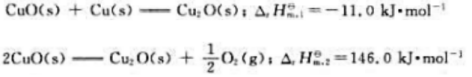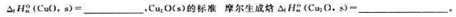题目内容
(请给出正确答案)
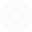
题目内容
(请给出正确答案)
已知下列两个热化学方程式:H2(g)+O2(g)==== H2O(l) ΔH =-285.8kJ/mol;X(g)+5O2(g)====3Y(g)+4H2O(l) ΔH =-2220.0kJ/mol,由实验测得:H2和X的混合气体,每5mol完全燃烧时可放出3837kJ热量,则此混合气体中,H2和X的体积比是()
A.1:3
B.3:1
C.1:4
D.1:1
 答案
答案
A、1:3
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案

 更多“已知下列两个热化学方程式:H2(g)+O2(g)==== H…”相关的问题
更多“已知下列两个热化学方程式:H2(g)+O2(g)==== H…”相关的问题