题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
Kyanite Pizzas Co (Kyanite) operates a large chain of fast food restaurants. You are an au
Kyanite Pizzas Co (Kyanite) operates a large chain of fast food restaurants. You are an audit supervisor of Jasper & Co and are currently preparing the audit programmes for the audit of Kyanite’s financial statements for the year ended 31 March 2016. You are reviewing the notes of last week’s meeting between the audit manager and finance director where two material issues were discussed.
(i) Property, plant and equipment
In the past Kyanite has received negative press reports over the condition of its fast food restaurants, with comments suggesting they are old fashioned and tired looking. Therefore during the year the company undertook a full review of all its assets and carried out extensive refurbishments to the majority of its restaurants. This review resulted in a significant amount of ageing fixtures and fittings being disposed of and a significant amount of capital expenditure was invested in all remaining restaurants. (6 marks)
(ii) Equity The refurbishment was financed via a share issue in April 2015 at a premium of $1·6 million. (4 marks)
Required:
Describe substantive procedures you should perform. to obtain sufficient and appropriate audit evidence in relation to the above two matters.
Note: The mark allocation is shown against each of the two matters above.
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案


 更多“Kyanite Pizzas Co (Kyanite) op…”相关的问题
更多“Kyanite Pizzas Co (Kyanite) op…”相关的问题
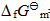 =-197.15kJ·mol-1,求尿素的合成反应在298.15K时的
=-197.15kJ·mol-1,求尿素的合成反应在298.15K时的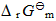 和KΘ。
和KΘ。 H2O(g)+CO(NH2)2(s)
H2O(g)+CO(NH2)2(s)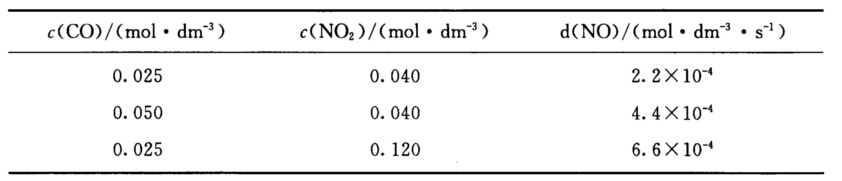 (1)写出反应的速率方程; (2)求650 K时的速率常数; (3)当c(CO)=0.10 mol.dm-3,c(NO2)=0.16 mol.dm-3时,求650 K时的反应速率; (4)若800 K时速率常数为23.0 dm3mol-1.s-1,求反应的活化能。
(1)写出反应的速率方程; (2)求650 K时的速率常数; (3)当c(CO)=0.10 mol.dm-3,c(NO2)=0.16 mol.dm-3时,求650 K时的反应速率; (4)若800 K时速率常数为23.0 dm3mol-1.s-1,求反应的活化能。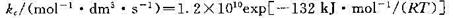
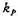 值;(2)反应进行10h后,NO的分压。
值;(2)反应进行10h后,NO的分压。